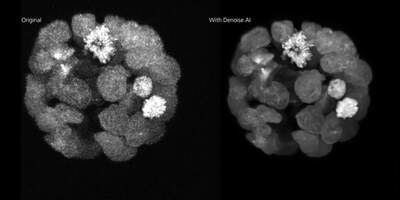
Immagine per gentile concessione di Laurence Pelletier Lab, LTRI
A1 HD25 / A1R HD25
Sistema di microscopia confocale
Discontinuo Replaced by AX / AX R with NSPARC
Un microscopio confocale che cattura immagini con un campo visivo di 25 mm, fino a due volte l'area dei tradizionali point scanner
La cattura di immagini di campioni di grandi dimensioni, come tessuti, organi e organismi interi, richiede l’estensione dell'area rilevabile e l’aumento della velocità di acquisizione dell'immagine. Il microscopio confocale A1 HD25 / A1R HD25 ha il campo visivo più ampio (25 mm) sul mercato, consentendo agli utenti di esplorare oltre i tradizionali confini dell'imaging confocale.