Measuring Spatial and Temporal Ca2+ Signals in Arabidopsis Plants
Abstract
Developmental and environmental cues induce Ca2+ fluctuations in plant cells. Stimulus-specific spatial-temporal Ca2+ patterns are sensed by cellular Ca2+ binding proteins that initiate Ca2+ signaling cascades. However, we still know little about how stimulus specific Ca2+ signals are generated. The specificity of a Ca2+ signal may be attributed to the sophisticated regulation of the activities of Ca2+ channels and/or transporters in response to a given stimulus. To identify these cellular components and understand their functions, it is crucial to use systems that allow a sensitive and robust recording of Ca2+ signals at both the tissue and cellular levels. Genetically encoded Ca2+ indicators that are targeted to different cellular compartments have provided a platform for live cell confocal imaging of cellular Ca2+ signals. Here we describe instructions for the use of two Ca2+ detection systems: aequorin based FAS (film adhesive seedlings) luminescence Ca2+ imaging and case12 based live cell confocal fluorescence Ca2+ imaging. Luminescence imaging using the FAS system provides a simple, robust and sensitive detection of spatial and temporal Ca2+ signals at the tissue level, while live cell confocal imaging using Case12 provides simultaneous detection of cytosolic and nuclear Ca2+ signals at a high resolution.
Introduction
The plant cell responds to the environment via signaling that coordinates cell actions. An early cell signaling event in response to environmental stimuli is a transient Ca2+ increase. The pattern, or signature of a transient increase in free Ca2+ concentration is characterized by its amplitude, frequency, and duration. Distinct spatio-temporal Ca2+ signatures regulate different cellular activities1. Specific stimuli, such as heat, cold, salt, drought, light, or plant hormones, may fine-tune the spatio-temporal activity of membrane-localized Ca2+ channels and/or transporters, resulting in specific Ca2+ signatures. Although Ca2+ transporters have been well characterized, little is known about the molecular identities and functions of Ca2+ channels in plants1. Genetic screens for mutants with altered Ca2+ response to stress stimuli may be an effective approach for identifying the components that compose Ca2+ signatures. Recently several Aequorin based Ca2+ detection systems have been developed that facilitate genetic screens for Ca2+ signaling components in response to pathogen attack and abiotic stress2-4.
Aequorin was first used to detect Ca2+ signals in plants in the early 1990s5. Since then, Aequorin has been targeted to different cellular compartments, such as the cytoplasm5, nucleus6, chloroplasts7, tonoplast8, mitochondria9, and stroma10, as well as to different cell types in the root to monitor cell specific Ca2+ signals11. Aequorin based Ca2+ measurements reveal the spatial and temporal Ca2+ response of a population of cells to stress stimuli. However, in most cases, the Ca2+ responses of single cells are unsynchronized in the responding tissue4. Therefore, Aequorin Ca2+ recording does not necessarily report the Ca2+ signal in individual cells. In recent years, genetically encoded fluorescent protein (FP)-based Ca2+ indicators, such as yellow cameleon (YCs)12 and CASEs1213 have been used to study Ca2+ signaling with high subcellular resolution. YCs are fluorescence resonance energy transfer (FRET)-based Ca2+ indicators, containing CFP and YFP variants linked by the Ca2+-binding protein calmodulin and calmodulin-binding peptide M13. Calmodulin undergoes a conformational change as it binds to Ca2+, thereby brings CFP and YFP closer together, resulting in increased energy transfer (enhanced FRET). The FRET level over time, calculated roughly as the ratio of YFP to CFP signal intensities, reflects intracellular Ca2+ dynamics. Several YC versions have been used in plants. YC3.6 was targeted to the cytosol14,15, nucleus16, mitochondria17, and plasma membrane18, and YC4.6 and D4ER were targeted to the ER15,19, and D3cpv was targeted to the peroxisomes20. Transgenic plants expressing YCs allow the live-cell imaging of Ca2+ dynamics within different cellular compartments of different cell types. CASEs (presumably Calcium sensor) are single circularly permuted fluorescent proteins (cpFPs) harboring a calmodulin and calmodulin-binding peptide M13. Upon binding to Ca2+, CASEs undergo conformational changes, leading to an increase of fluorescence intensity. The correlation between the CASE’s fluorescence response and Ca2+concentration allows intracellular Ca2+ dynamics to be measured quantitatively. The Case12 variant has 12 fold increased fluorescence in the Ca2+-saturated forms. N. benthiminana plants transiently expressing Case12 or Arabidopsis plants stably expressing Case 12 were used to study Ca2+ signaling in defense and abiotic stress4,21 . Asynchronous spatial and temporal Ca2+ oscillations in cells responding to pathogen attack, or to dehydration stress have been revealed with Case12 based Ca2+ imaging.
Here, we present detailed instructions for Aequorin based luminescence imaging of tissue- and stimuli specific Ca2+ dynamics in Arabidopsis seedlings, and for confocal imaging of cytosolic and nuclear Ca2+ dynamics in Arabidopsis root cells that express Case 12. Luminescence imaging of FAS could be adapted to analyze stress-induced Ca2+ dynamics in intact plants or tissues not described here, or to screen mutagenized Arabidopsis plant populations for mutants with altered stress induced Ca2+ signals. The live cell Ca2+ imaging setup could be adapted to analyze Ca2+ dynamics within different subcelluar compartments or in different cell types using other Ca2+ indicators.
Protocol
1. Aequorin Based Ca2+ Imaging Using the FAS System
- Prepare seedlings for luminescence imaging. Sterilize seeds of Arabidopsis plants expressing Aequorin with 10% bleach solution containing 0.01% Triton-100. Sow the sterile seeds on a square plate (10 x 10 cm square Petri dish with grid,) containing full strength MS (Murashige and Skoog Basal Salt Mixture), 1% sucrose, and 1.2% agar. Place plates vertically in a growth chamber after stratification at 4 °C for 2 days (Figure 1A).
- Transfer the seedlings onto a film. Place an adhesive film (Figure 1B) on the top of 7-10 day old seedlings growing on the plate. Gently push the film by hand to ensure that seedlings adhere to the film (Figure 1C). Peel the film gently so that the seedlings remain adhered to the film (Figure 1D and 1E)
- Incubate the seedlings with cofactor. Place the adhered seedlings onto the square plate (10 x 10 cm) containing 15 ml of 2 μg/ml h-CTZ (coelenterazine) in water. Incubate the seedlings at room temperature for 4 hr to overnight (Figure 1F).
- Prepare for luminescence imaging. Take the film out of the h-CTZ solution and cut it down the middle, forming two pieces. Place each piece of film with seedlings face up in two different plates. Leave the plates in the dark for 5 min.
- Acquire luminescence images. In the dark, place the two plates next to each other on the stage of the luminescence imaging system (Figure 1G). Acquire images immediately upon adding 20 ml of stimuli solution to the plates simultaneously.
- Analyze luminescence images. Choose the same display range for all luminescence images. Crop the region of interest (ROI) and generate the images as JPEG files (Figure 2A). Alternatively, export the images as SPE format files and import them into the ImageJ image analysis software. Set measurements for calculation of the mean gray value of ROI. Select the same size of ROI area and measure the mean gray and present data as bar graphs (Figure 2B).
2. Live Cell Confocal Ca2+ Imaging
- Prepare seedlings for confocal imaging. Sterilize seeds of Arabidopsis plants expressing case12 with 10% bleach solution containing 0.01% Triton. Sow the sterile seeds on a plate containing full- strength MS salts, 1% sucrose, and 1.2% agar. Place plates vertically in a growth chamber after stratification at 4 °C for 2 days.
- Setup Imaging Chambers
- Assemble an imaging chamber using a slide and coverslip (Chamber A). Place a piece of water soaked cotton wool onto the middle of the slide. Then transfer one or two 5 day old seedlings from the plate onto the top of water soaked cotton wool. Stick small pieces of clay to each corner of a coverslip, and place coverslip on the top of seedlings to create a gap (chamber) between the coverslip and slide (Figure 3A, left panel). Connect one end of polyethylene tube (0.58 mm diameter) to a 1 ml syringe and place the other end of tube immediately adjacent to the chamber. Hold the tubing in place with tape (Figure 3A, right panel).
- Assemble an imaging chamber using a plexiglass chamber (Chamber B).
- Spread a thin layer of silicone grease around two polyethylene tubes (0.58 mm diameter) and press the coated tubes into the channels on each side of the plexiglass chamber (Figure 3B left panel). Spread a thin layer of silicone grease onto the surface of the chamber that contains the tube grooves and press a coverslip into the grease to seal one side of the chamber opening (cut out).
- Transfer a five day old seedling from the plate into the chamber and place a piece of cotton wool soaked with water on the top of the seedling. Spread silicone grease onto the other surface of the chamber, and press another coverslip on top to seal. Connect the end of one of the tubes to a 1 ml syringe. Leave the end of the other tube open (Figure 3B right panel).
- Prepare an imaging chamber using a chambered cover glass (Chamber C). Sterilize the chambered cover glass with 70% ethanol and leave it on the hood until dry. Add 0.6 ml or 0.2 ml of full strength MS medium containing 1% sucrose and 0.5% phytagel into each well of an 2-well chamber or an 8-well chamber (Figure 3C right panel), respectively. Sterilize seeds and sow the seeds directly in a chambered cover glass containing a thin layer of clear gel and let them grow vertically for 5 days (Figure 3C).
- Acquire images using a confocal microscope. To apply stress stimuli, such as salt, cold or peroxide, slowly inject about 200 μl of 150 mM NaCl, ice-cold water or 1 mM H2O2 solution into the chamber (Chamber A or B) just prior to acquiring images, or gently add 500 μl or 100 μl of stimulus solution to the well of a 2- or 8-well chamber, respectively. Capture images immediately after applying the stimulus solution using an inverted Nikon A1R confocal laser-scanning microscope with a 20X water immersion lens (numerical aperture 0.75). Collect a time series of images at 4 sec intervals with excitation and emission wavelengths of 488 nm and 500 - 550 nm, respectively, and at a pixel resolution of 512 x 512.
- Image Analysis
Using Nikon NIS-Elements, ROIs were drawn around each cell (or area) of interest. Total intensity within each ROI was measured over time using the Time Measurement dialog box (ImageJ could be used instead). Total intensity measurements were exported and processed in DataGraph. Ca2+ spike amplitude was defined as peak intensity minus resting intensity, duration as the time between initiation and completion of a spike, and period as the time interval between adjacent spike peaks of two spikes. t-test was used to compare the means.
Representative Results
Mannitol, NaCl and H2O2 were used as proxies for dehydration, salt and oxidative stress stimuli, respectively. To check if the heavy metal ion Cu2+ synergizes with any of these three stress stimuli, we compared the Ca2+response to each stimuli in the presence or absence of Cu2+. As shown in Figure 2, FAS luminescence imaging revealed that Arabidoposis seedlings responded differently to dehydration, salt and oxidative stress. For the concentration of stimuli we examined in this study, 75 mM NaCl induced the strongest Ca2+ response in leaves and roots during the first 40 sec of exposure (Figure 2A middle panel and Figure 2B), while 1 mM H2O2 induced a strong Ca2+ response both in leaves and roots during the first and second 40 sec (Figure 2A right panel). The solution of 400 mM mannitol only induced Ca2+ response in roots during the first 40s and very weak signals in leaves during the second 40 sec. Cu2+ strongly enhanced the amplitudes and duration of Ca2+ signals triggered by NaCl and mannitol (Figure 2A left panel). It appears that Cu2+ had inhibitory effects on H2O2 induced Ca2+ signals, reducing both the amplitude and duration (Figure 2A right panel and 2B).
Using Case12 expressing Arabidopsis plants, we measured Ca2+ dynamics in the cytosol and nuclei simultaneously. Molecular weight of Case12 is 46 kDa, therefore transgenic plants expressing Case12 have GFP signals in both the cytosol and nuclei of leaf and root cells (Figure 4A). Customized (Figure 3A and 3B) or commercial chambers (Figure 3C) were used for live cell imaging with a confocal microscope. Applying 150 mM NaCl to the seedling in the chamber, we observed changes of fluorescence intensity in both the cytosol and nucleus of the individual cells (Figure 4B and C; Supplementary Movie 1). Using NIS-Elements (Imaging software, Nikon Instruments Inc.), we measured the fluorescence intensity in regions of interest (ROI) over time. The amplitude, duration and frequency of cytosolic and nuclear Ca2+ oscillation are presented graphically in Figure 4B. The amplitude and period of Ca2+ spikes in the cytoplasm and the nuclei vary greatly between cells of the same root, as well as across cells in different roots. The amplitude and period of cytosolic Ca2+ spikes were 60.36 ± 45.22 AU (arbitrary unit) and 58.24 ± 15.70 sec, respectively. The amplitude and period of nuclear Ca2+ spikes were 316.26 ± 75.24 (AU) and 61.71 ± 16.31 sec, respectively. In contrast, the duration of cytosolic and nuclear Ca2+ spikes were similar between cells, which were 17.43 ± 3.67 sec and 17.33 ± 2 sec, respectively. The amplitude of Ca2+ spikes decreased over time, which may be due to the change of the focal plane and/ or a loss of response to stress stimuli because of an unfavorable in vitro condition. These results show that salt stress triggers Ca2+ oscillation in both the cytoplasm and nucleus.

Figure 1. Aequorin based FAS system for measuring spatial-temporal Ca2+ dynamics in response to stress stimuli. A) Grow Arabidopsis seedlings vertically on a plate containing MS media. B, C) Place an adhesive film (B) on the top of seedlings (C). D, E) Transfer 10 day old seedlings onto the adhesive film.F) Incubate seedlings adhered to the film with 2 µg/ml of h-CTZ in water for 4 hr. G) Cut the film down the middle and place each piece into a different empty plate. Leave the plates in the dark for 5 min. Apply simultaneously control and stimuli solutions (be sure the seedlings are entirely covered), and acquire images immediately. The scale bar is 5 cm.
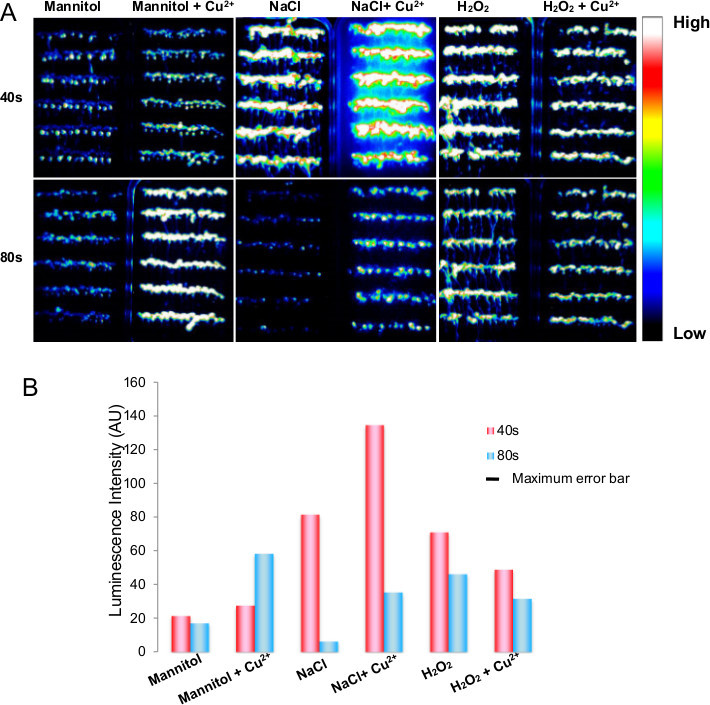
Figure 2. Comparison of spatial-temporal Ca2+ response of 10 day seedlings to different stress stimuli. A) A time series of luminescence images of seedlings subjected to 400 mM mannitol and 400 mM mannitol plus 1 mM CuCl2 (A, left panel), 75 mM NaCl and 75 mM NaCl plus 1 mM CuCl2 (A, middle panel), and 1 mM H2O2 or 1 mM H2O2 plus 1 mM CuCl2 (A, right panel). Upper and lower panels of A are luminescence images acquired during first 40 sec and second 40 sec, respectively. An intensity scale bar indicates an increase of luminescence signals from low (black) to high (white). B) Bar chart of average luminescence intensity of seedlings in response to the indicated stress stimuli.

Figure 3. Setup of a live cell confocal imaging experiment. A) A customized chamber is built with a slide, coverslip with four pieces of clay and cotton wool. Two 5 day old seedlings are placed on the slide. One end of a polyethylene tube is placed next to the assembled chamber and fixed with a pieces of tape while the other end is connected to a 1 ml syringe. B) A customized slide made with plexiglass has a open chamber in the middle and two channels that are connected to the two sides of channel. One coverslip is placed on the top of the slide and two polyethlene tubes are placed in the channel. Coverslip and tubes are fixed in the position with silicone grease. The end of one of the tubes is connected to a 1 ml syringe and the end of another tube remains open. A 5 day old seedling is placed in the chamber and a coveslip is put on the top of the seedling and sealed with silicone grease. The plexigalss slide with the assembled chamber is placed on the stage of the microscope. C) Arabidopsis seedlings are grown in two wells (left) or 8 wells (right) of the chambered cover glass containing a thin layer of 0.6% of phytagel in MS media.
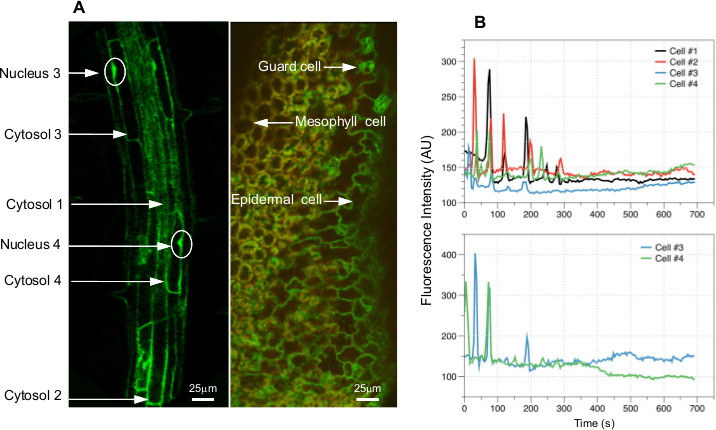
Figure 4. Cytosolic and nuclear Ca2+ oscillation in response to salt stress. A) Confocal images show that Case12 is expressed in the cytoplasm and nuclei of root cells (left panel) and leaf cells (right panel) of transgenic plants expressing Case12. The image on the right panel is a merged image of red autofluorescence of the chloroplasts and green fluorescence of Case12. B) Salt stress (150 mM NaCl) induced Ca2+ oscillation in the cytosol (upper panel) and nuclei (lower panel) of root cells. Locations of the measured cells are indicated by arrows with numbers on panel.
Discussion
We have demonstrated a FAS system for recording the spatial-temporal Ca2+ response of Arabidopsis seedlings. This FAS Ca2+ recording system provides a simple, sensitive and robust approach that could be adapted for measuring Ca2+ dynamics triggered by various stimuli in addition to the abiotic stress stimuli that are presented here. Using this system, we can easily compare tissue- or stimuli-specific spatial-temporal Ca2+ dynamics at the whole plant level. The high sensitivity of FAS allows the use of low intensity stimuli for examining Ca2+ responses, which is important for avoiding artifacts caused by physical damages to cells when high intensity stress is used. Seven to 10 day old Arabidopsis seedlings showed tissue or stimuli-specific Ca2+response, though seedlings at different physiological stages may have different sensitivity and specificity in response to a given stimulus. Therefore it is preferable to use 7-10 day old seedlings to increase the throughput of FAS measurements, in which around 100 seedlings could be adhered onto one piece of film. Together with easy and simple performance, high sensitivity and reproducibility, the FAS system could also be adapted for screening EMS mutagenized mutants for altered Ca2+ responses. For screening purposes, it is crucial to have uniform seedlings that are well grown on the plate and adhered onto the film. Four to five hours incubation of film adhered seedlings with h-CTZ is usually sufficient for reconstitution of aequorin. Using the adhesive film with holes is preferable because reaction of apoaequorin and cofactor requires oxygen. For the comparison of tissue or stimuli-specific Ca2+ responses, using groups of seedlings originally from the same film is critical because the state of seedlings and/or the reconstitution condition may affect the total amount of functional aequorin. When these requirements are met, the difference of luminescence response is proportional to the tissue- or stimulus-specific Ca2+ response.
Aequorin based measurement of Ca2+ signals reflect the response of a population of the cells. However, considering the heterogeneity of cell types and/or the different accessibility of cells in the tissues to the stimuli, the results of Aequorin based Ca2+ measurements do not equate to the behavior of any one cell. Understanding Ca2+ dynamics at the cellular level requires a Ca2+ indicator that can be detected with subcellular resolution. Live cell confocal imaging of fluorescence protein (FP)-based Ca2+ indicators provides an advanced Ca2+ detection system for recording cellular Ca2+ dynamics with cellular resolution in a desired tissue. Here, using live cell confocal imaging and FP-based Ca2+ indicators, we recorded subcellular Ca2+ transients in both the cytoplasm and nuclei simultaneously. Two types of FP-based Ca2+ indicators have been implemented in plants with different characteristics. Compared to YCs, single GFP-based Ca2+ indicator has some advantages: 1. Case12 does not require YFP and CFP filter sets or the many assumptions and complex calculations needed to measure FRET22. Case12 has single excitation/emission maxima and can be easily detected using standard GFP filter set. 2. Case12 is stable under physiological pH, and is a relatively a small protein (46 kDa). When expressed in plants, non-targeted Case12 appears in the cytoplasm as well as the nuclei, so Case12 transgenic plants can be used to track cytosolic and nuclear Ca2+ dynamics simultaneously. A drawback of this reporter is that it is a non-ratiometric indicator, so fluorescence levels are also affected by factors that are unrelated to Ca2+ concentration, such as the expression level of Case12 and local pH Nevertheless, Case12 is adequate to determine cellular Ca2+ concentration for the comparison of spatial-temporal Ca2+ dynamics in the cytoplasm and organelles between materials with different genetic backgrounds under the same experimental conditions. In the future, new GFP based Ca2+ indicators with higher signal-to-noise ratio, fast kinetics and greater response, like GCaMP324 and GCaMP525 demonstrated in animals, could be better indicators for recording cellular or subcelluar Ca2+ dynamics in plants.
The Aequorin and Case12 detection systems described here provide nondestructive in vivo approaches for the measurement of spatial-temporal Ca2+ dynamics at the whole seedling and subcellular levels, respectively. In both systems, the physiological state or growth conditions could affect the Ca2+ responses of the seedlings or cells. If physically damaged or not grown in a good condition, cells could have weak or even no Ca2+ responses. In addition, the duration of spatial and temporal Ca2+ signals are also affected by the conditions of the seedling during the measurements. The luminescence or fluorescence signals may decrease over time due to stimuli-induced damage, and/or the laser-induced photo-damage in the case of confocal imaging. Although stimulus-induced damage cannot be avoided, phototoxicity can be reduced by limiting the intensity and/or frequency of laser exposure. In addition, Aequorin based Ca2+ recording system requires a reconstitution step. Factors that interrupt the diffusion of CTZ to the cell and/or reaction of apoaequorin with CTZ would affect luminescence signals that are related directly to Ca2+ concentration. Therefore the concentration of CTZ, as well as the duration and conditions of CTZ incubation need to be optimized for best sensitivity and reproducibility. Genetically encoded Ca2+ indicators may inaccurately report cellular Ca2+ concentrations for other reasons, including unintended targeting to specific subcellular compartments, temperature sensitivity, or interference of the reporter with the cellular Ca2+ signaling machinery23. We found that Aequorin or Case12 expressing plants have no obvious morphological or conditional phenotypes such as altered stress tolerance. Therefore it is unlikely that these genetically encoded Ca2+ indicators have major interferences with Ca2+ signaling in plants.
In summary, we present here detailed instructions for the use of two Ca2+ detection systems for measuring spatial and temporal Ca2+ signals at both the whole plant seedling and subcellular levels. These two systems can be used to determine tissue (or cell type) or stimuli-specific Ca2+ dynamic patterns with high sensitivity and reproducibility. Therefore they are powerful tools for elucidating the gene networks that underlie Ca2+ signaling in response to various environmental cues.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful to B. Stevenson for technical assistance and Dr Marc R. Knight for providing Aequorin transgenic line. This work was funded by the National Institutes of Health Grant R01 GM059138.
Materials
| Name | Company | Catalog Number | Comments |
| 10 cm x 10 cm square Petri dish | VWR | 60872-310 | |
| Adhesive film | VWR | 60941-062 | |
| Polyethylene tubing | PerkinElmer | 9908265 | |
| 1 ml syringe | VWR | 53548-000 | |
| Silicone grease | Beckman | 335148 | |
| 2-well chambered cover glass | Nalge Nunc international | 155379 | |
| 8-well chambered cover glass | Nalge Nunc international | 155409 | |
| Luminescence imaging system | Princeton Instruments | N/A | |
| Inverted confocal laser-scanning microscope | Nikon Instruments Inc. | N/A | Nikon A1R |
| Imaging software | Nikon Instruments Inc. | N/A | Nikon NIS-Elements |
| DataGraph | Visual Data Tools Inc | N/A | DataGraph 3.1.1 is the newest version |
| Coelenterazine | NanoLight Technolgies | #301B NF-BCTZ-FB | |
| All purpose bleach | Any local store | N/A | |
| Triton X-100 | Fisher | BP151500 | |
| MS salt | Phytotechnology Labs | M524-50L | |
| Sucrose | VWR | BDH8029-12KG | |
| Agar | Sigma | A1296-5KG | |
| Phytagel | Sigma | P8169-1KG |
References
- Kudla, J., Batistic, O., Hashimoto, K. Calcium signals: the lead currency of plant information processing.Plant Cell. 22(3), 541-63 doi: 10.1105/tpc.109.072686 (2010).
- Pan, Z., Zhao, Y., Zheng, Y., Liu, J., Jiang, X., & Guo, Y. A high-throughput method for screening Arabidopsis mutants with disordered abiotic stress-induced calcium signal. J. Genet. Genomics. 39(5), 225–235 doi:10.1016/j.jgg.2012.04.002 (2012).
- Ranf, S. et al. Defense-related calcium signaling mutants uncovered via a quantitative high-throughput screen in Arabidopsis thaliana. Mol. Plant. 5(1), 115–130 doi:10.1093/mp/ssr064 (2012).
- Zhu, X., Feng, Y., Liang, G., Liu, N., & Zhu, J. K. Aequorin-based luminescence imaging reveals stimulus- and tissue-specific Ca2+ dynamics in Arabidopsis plants. Mol. Plant. 6(2), 444-455 doi:10.1093/mp/sst013 (2013).
- Knight, M. R., Campbell, A. K., Smith, S. M., & Trewavas, A. J. Transgenic plant aequorin reports the effects of touch and cold-shock and elicitors on cytoplasmic calcium. Nature. 352(6335), 524–526 doi:10.1038/352524a0 (1991).
- Van der Luit A. H., Olivari, C., Haley, A., Knight, M. R., & Trewavas, A. J. Distinct calcium signalling pathways regulate calmodulin gene expression in tobacco. Plant Physiol. 121(3), 705–714 doi:10.1104/pp.121.3.705 (1999).
- Johnson, C. H. et al. Circadian oscillations of cytosolic and chloroplastic free calcium in plants. Science.269(5232), 1863–1865 doi:10.1126/science.7569925 (1995).
- Knight, H., Trewavas, A. J., & Knight, M. R. Cold calcium signaling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell. 8(3), 489–503 doi:10.1105/tpc.8.3.489 (1996).
- Logan, D. C., & Knight, M. R. Mitochondrial and cytosolic calcium dynamics are differentially regulated in plants. Plant Physiol. 133(1), 21-24 doi:10.1104/pp.103.026047 (2003).
- Mehlmer, N., Parvin, N., Hurst, C. H., Knight, M. R., Teige, M., & Vothknecht, U.C. A toolset of aequorin expression vectors for in planta studies of subcellular calcium concentrations in Arabidopsis thaliana. J. Exp. Bot. 63(4), 1751-1761 doi: 10.1093/jxb/err406 (2012).
- Kiegle, E., Moore, C. A., Haseloff, J., Tester, M. A., & Knight, M. R. Cell-type-specific calcium responses to drought, salt and cold in the Arabidopsis root. Plant J. 23(2), 267-278 doi:10.1046/j.1365-313x.2000.00786.x (2000).
- Palmer, A. E., & Tsien, R.Y. Measuring calcium signaling using genetically targetable fluorescent indicators.Nat. Protoc. 1(3), 1057–1065 doi:10.1038/nprot.2006.172 (2006).
- Souslova, E. et al. Single fluorescent protein-based Ca2+ sensors with increased dynamic range. BMC. Biotechnol. 7, 37-46 doi:10.1186/1472-6750-7-37 (2007).
- Tanaka, K., Swanson, S., Gilroy, S., & Stacey, G. Extracellular nucleotides elicit cytosolic free calcium oscillations in Arabidopsis. Plant Physiol. 154(2), 705–719 doi:10.1104/pp.110.162503 (2010).
- Iwano, M. et al. Fine-tuning of the cytoplasmic Ca2+ concentration is essential for pollen tube growth. Plant Physiol. 150(3), 1322–1334 doi:10.1104/pp.109.139329 (2009).
- Sieberer, B., Chabaud. M., Timmers, A., Monin, A., Fournier, J., & Barker, D. A nuclear-targeted Cameleon demonstrates intranuclear Ca2+ spiking in Medicago truncatula root hairs in response to rhizobial nodulation factors. Plant Physiol. 151(3), 1197–1206 doi:10.1104/pp.109.142851 (2009).
- Loro, G., Drago, I., Pozzan, T., Lo Schiavo, F., Zottini, M., & Costa, A. Targeting of Cameleons to different subcellular compartments reveals a strict cytoplasmic/mitochondrial Ca2+ handling relationship in plant cells. Plant J. 71(1), 1–13 doi:10.1111/j.1365-313X.2012.04968.x (2012).
- Krebs, M. et al. FRET-based genetically encoded sensors allow high-resolution live cell imaging of Ca2+ dynamics. Plant J. 69(1), 181–192 doi:10.1111/j.1365-313X.2011.04780.x (2012).
- Bonza, M. C., Loro, G., Behera, S., Wong, A., Kudla, J., & Costa, A. Analyses of Ca2+ accumulation and dynamics in the endoplasmic reticulum of Arabidopsis root cells using a genetically encoded Cameleon sensor. Plant Physiol. 163(3), 1230-1241 doi:10.1104/pp.113.226050 (2013).
- Costa, A. et al. H2O2 in plant peroxisomes: An in vivo analysis uncovers a Ca2+ dependent scavenging system. Plant J. 62(5), 760–772 doi:10.1111/j.1365-313X.2010.04190.x (2010).
- Zhu, X., Caplan, J., Mamillapalli, P., Czymmek, K., & Dinesh-Kumar, S. P. Function of endoplasmic reticulum calcium ATPase in innate immunity-mediated programmed cell death. EMBO J. 29(5), 1007-1018 doi:10.1038/emboj.2009.402 (2010).
- Roszik, J., Lisboa, D., Szöllosi, J., & Vereb, G. Evaluation of intensity-based ratiometric FRET in image cytometry--approaches and a software solution. Cytometry A. 75(9), 761-767 doi:10.1002/cyto.a.20747 (2009).
- Kotlikoff, M. I. Genetically encoded Ca2+ indicators: using genetics and molecular design to understand complex physiology. J. Physiol. 578(Pt 1), 55–67 doi:10.1113/jphysiol.2006.120212 (2007).
- Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat methods. 6(12), 875–881. doi: 10.1038/nmeth.1398 (2009).
- Akerboom, J. et al. Optimization of a GCaMP calcium indicator for neural activity imaging. J Neurosci.32(40), 13819–13840 (2012).
Note: The institutions and job titles listed with each researcher reflect their affiliation at the time of the interview.